Applications and Challenges of Chromatography Resins in Large-Scale Manufacturing
In large-scale manufacturing, chromatography resins are not only the core of downstream purification processes, but also a critical factor in cost control and quality assurance. How can the right resin be selected in a scientific and rational way? This guide takes you straight to the key considerations and addresses the real challenges faced by the industry.
Balancing Efficiency and Cost: Choosing the Right Resin Determines Success
In biopharmaceutical manufacturing, medical aesthetics, and the extraction of high-value food ingredients, chromatography technology functions as the “heart” of production, with chromatography resins representing its most critical component. As processes transition from laboratory scale to industrial manufacturing, the rational selection of chromatography resins becomes a decisive factor for success, directly impacting product quality, production costs, and supply chain security.
At industrial production scale, the increase in column diameter leads to the loss of wall-support effects, while higher volumetric flow rates result in reduced bed porosity. As a consequence, strong physical and chemical stability becomes the foundation for successful scale-up of chromatography resins. Before selecting a chromatography resin, key factors such as scale-up performance, lot-to-lot consistency, pressure–flow characteristics, and resin lifetime should be thoroughly evaluated.
Physical Properties: Balancing Pressure and Separation Efficiency
In large-scale chromatography operations, pressure control represents the primary challenge. Conventional agarose-based resins typically tolerate operating pressures below 0.3 MPa; exceeding this limit can lead to resin compression and deformation, ligand detachment, and in severe cases, mesh weld failure or even column rupture. For large-diameter chromatography columns ranging from 1.2 to 2.0 m, these risks are further amplified.
With many years of focus on chromatography resin development, Bestchrom has addressed the inherent softness of conventional agarose-based matrices through the development of its Diamond base matrix platform. This innovation effectively resolves common scale-up challenges faced by customers, including excessive back pressure, limited achievable flow rates, and bed collapse during large-scale production. Currently, Bestchrom offers a high-rigidity agarose base matrix with a D50 (median particle size) of 40 µm (Diamond Mustang), as well as a range of high-rigidity agarose-based resins with D50 (median particle size) values of 60, 75, 80, and 90 µm, providing greater flexibility for both capture and polishing applications.
Extrem A Diamond, an affinity resin developed by Bestchrom, is a high-rigidity medium with a D50 (median particle size) of 60 µm. The pressure–flow performance was evaluated in a chromatography column with an inner diameter of 800 mm and a bed height of 25.5 cm, as shown in Fig. 1. At a system linear velocity of 300 cm/h, the column inlet pressure was only 1.66 bar. No bed collapse or water layer formation was observed, demonstrating excellent pressure resistance and providing strong assurance for industrial-scale process scale-up.
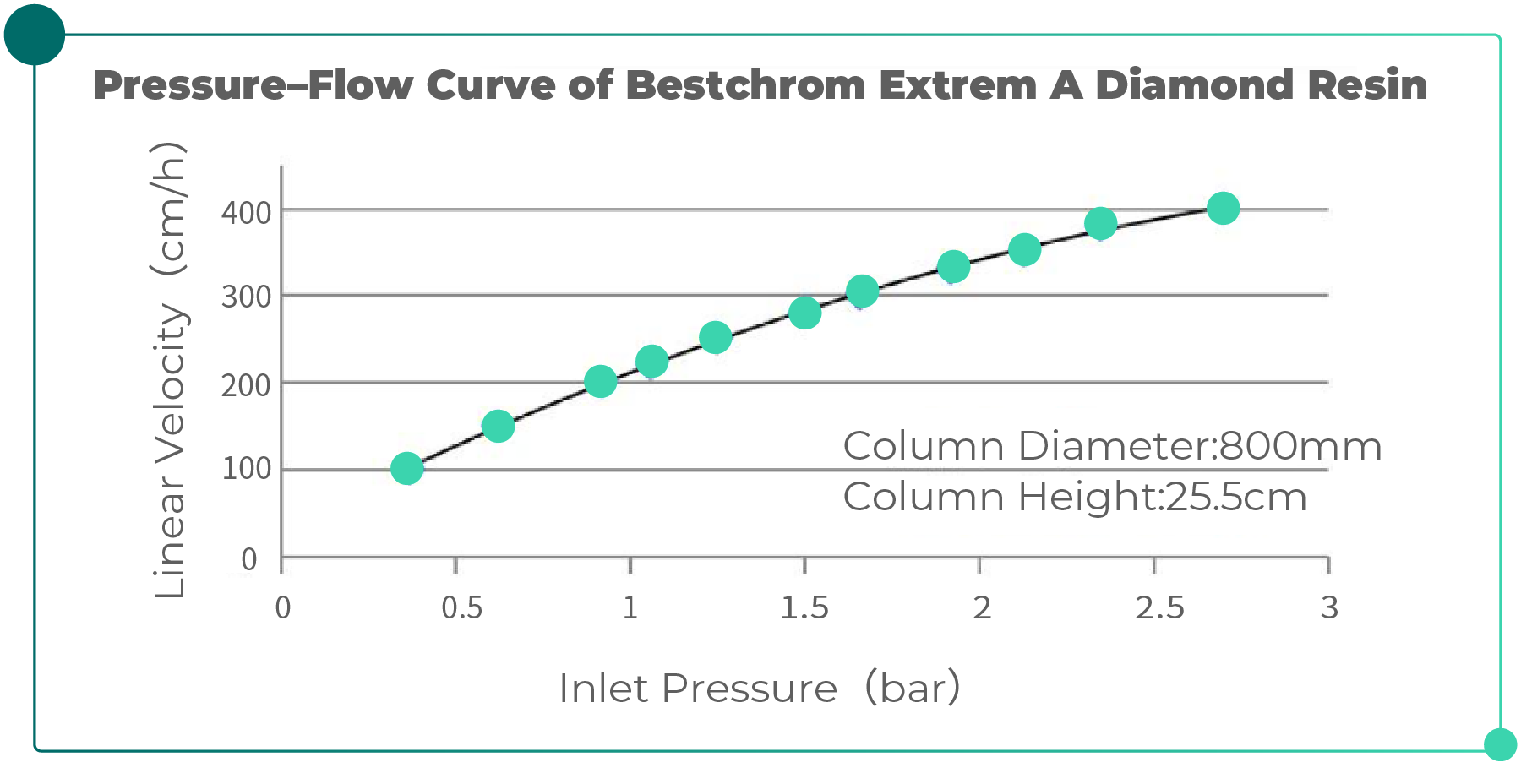
Fig. 1. Pressure–Flow Curve of Bestchrom Extrem A Diamond Resin
Long Service Life: The Key to Reducing Total Cost of Ownership (TCO)
When evaluating resin costs, the initial purchase price alone should not be the sole consideration. Instead, the cost per gram of purified product over the entire service lifetime of the resin should be assessed. Resin lifetime—defined by the number of reusable cycles—is a critical parameter. For example, a resin that can be reused for more than 100 cycles is significantly more cost-effective than a lower-priced resin limited to only 20 cycles. Therefore, comprehensive lifetime validation studies (cycle studies) are an essential prerequisite for large-scale manufacturing, ensuring that the resin maintains stable performance after repeated CIP/SIP procedures. The figure below shows the results of a 0.1 M alkaline soaking test performed on Bestchrom affinity resins, demonstrating their excellent alkaline stability.
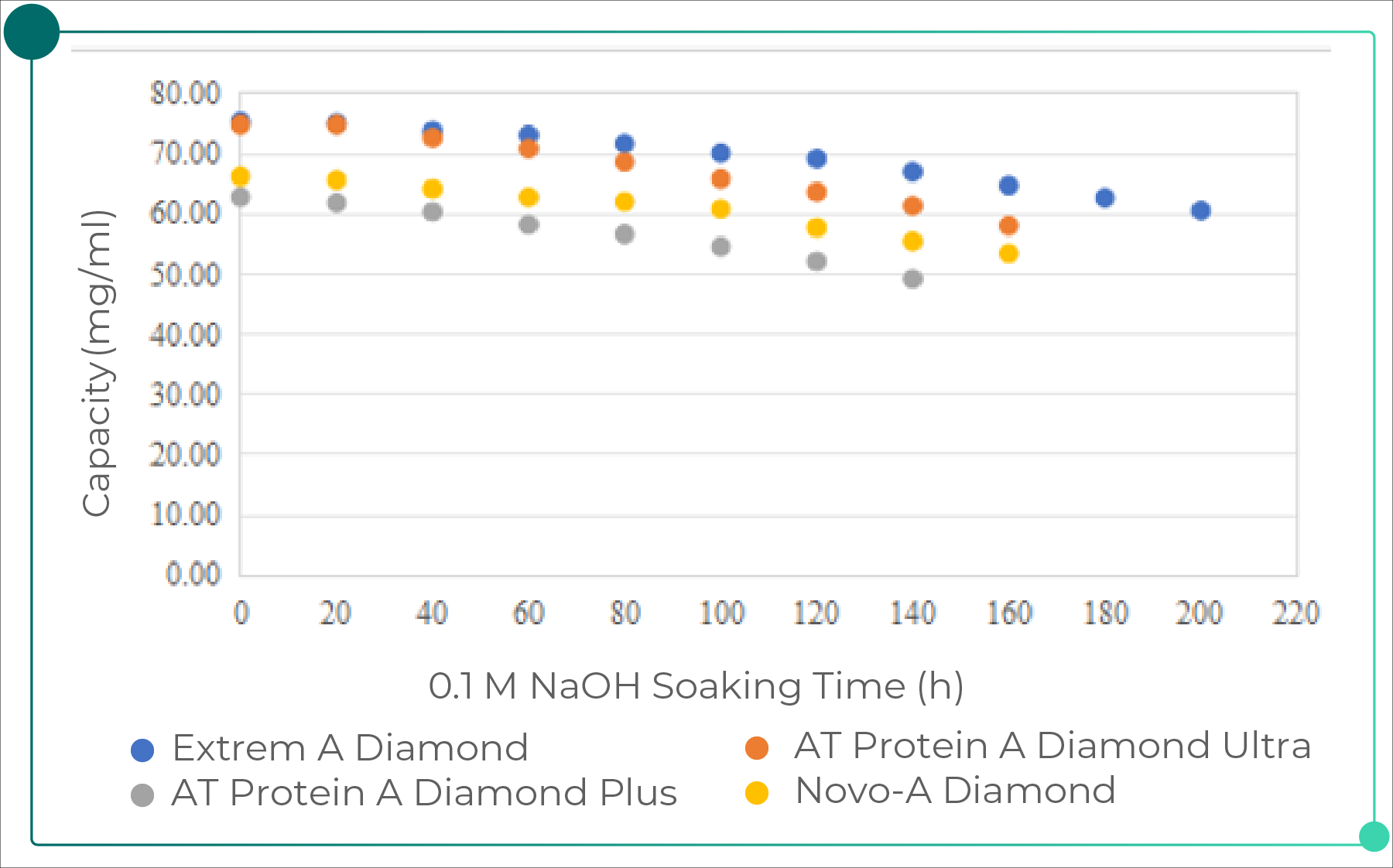
Fig. 2. Results of 0.1 M Alkaline Soaking Test for Bestchrom Affinity Resins
Process Scale-Up and Consistency: Ensuring Stable Manufacturing Performance
◉ Challenges
During scale-up, issues such as reduced separation performance and decreased binding capacity may arise. Lot-to-lot consistency of the resin is therefore critical, as even minor variations can lead to process instability and, in some cases, compromise product quality.
◉ Mitigation Strategies
• Early implementation of Quality by Design (QbD):
Conduct comprehensive design space exploration during early process development to understand the impact of critical process parameters (CPPs) on critical quality attributes (CQAs), thereby providing sufficient process flexibility and a sound scientific basis for scale-up.
• Rigorous supplier qualification and auditing:
Ensure that resin suppliers maintain rigorous QC standards and quality management systems (e.g., ISO 9001), and demonstrate stable lot-to-lot consistency (e.g., RSD < 5%).
• Comprehensive scale-up studies:
Perform thorough validation at the pilot scale to confirm the reliability of scale-up models prior to full-scale manufacturing.
The figure below presents the results of multi-lot testing of Bestchrom chromatography resins, demonstrating relatively stable lot-to-lot production consistency.
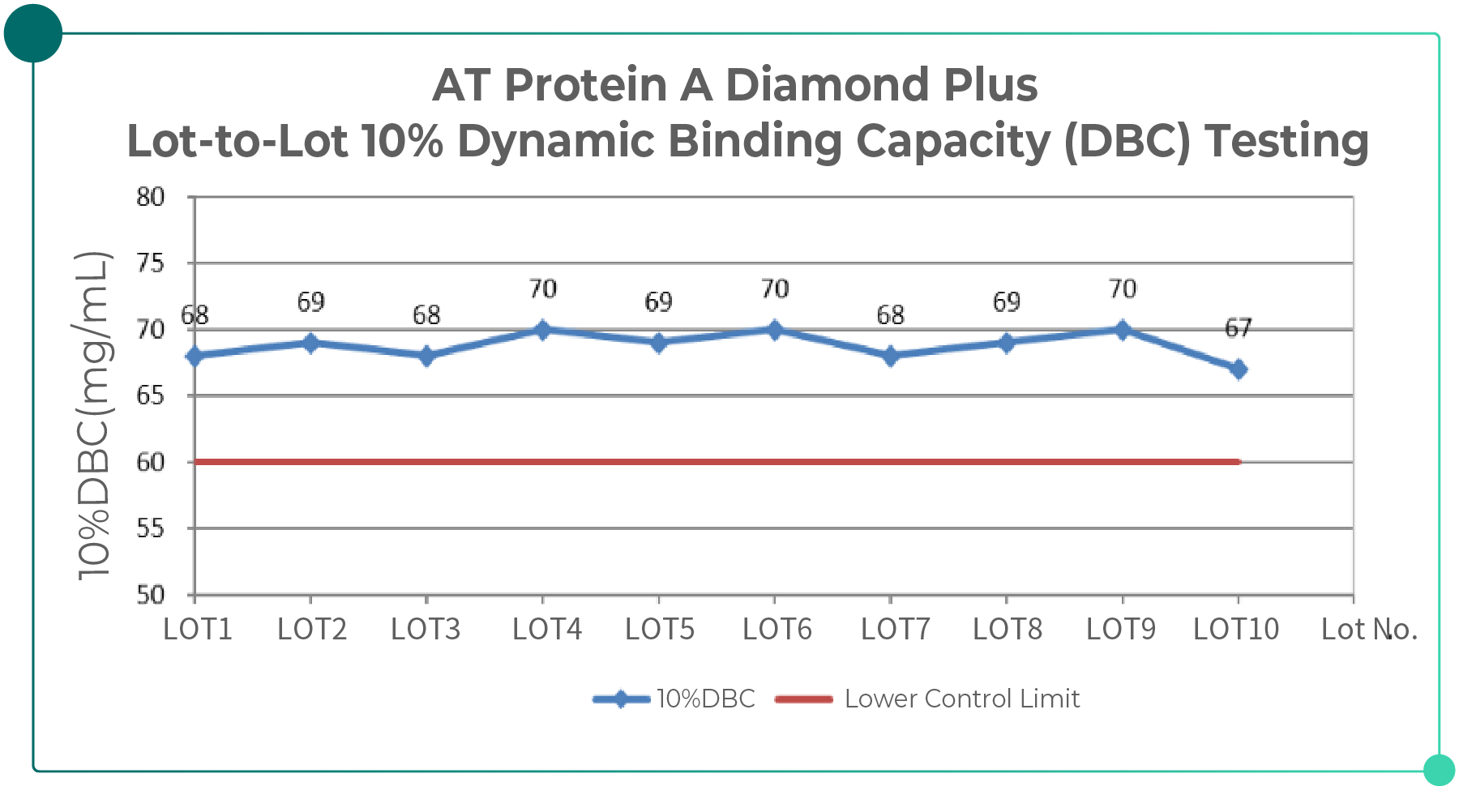
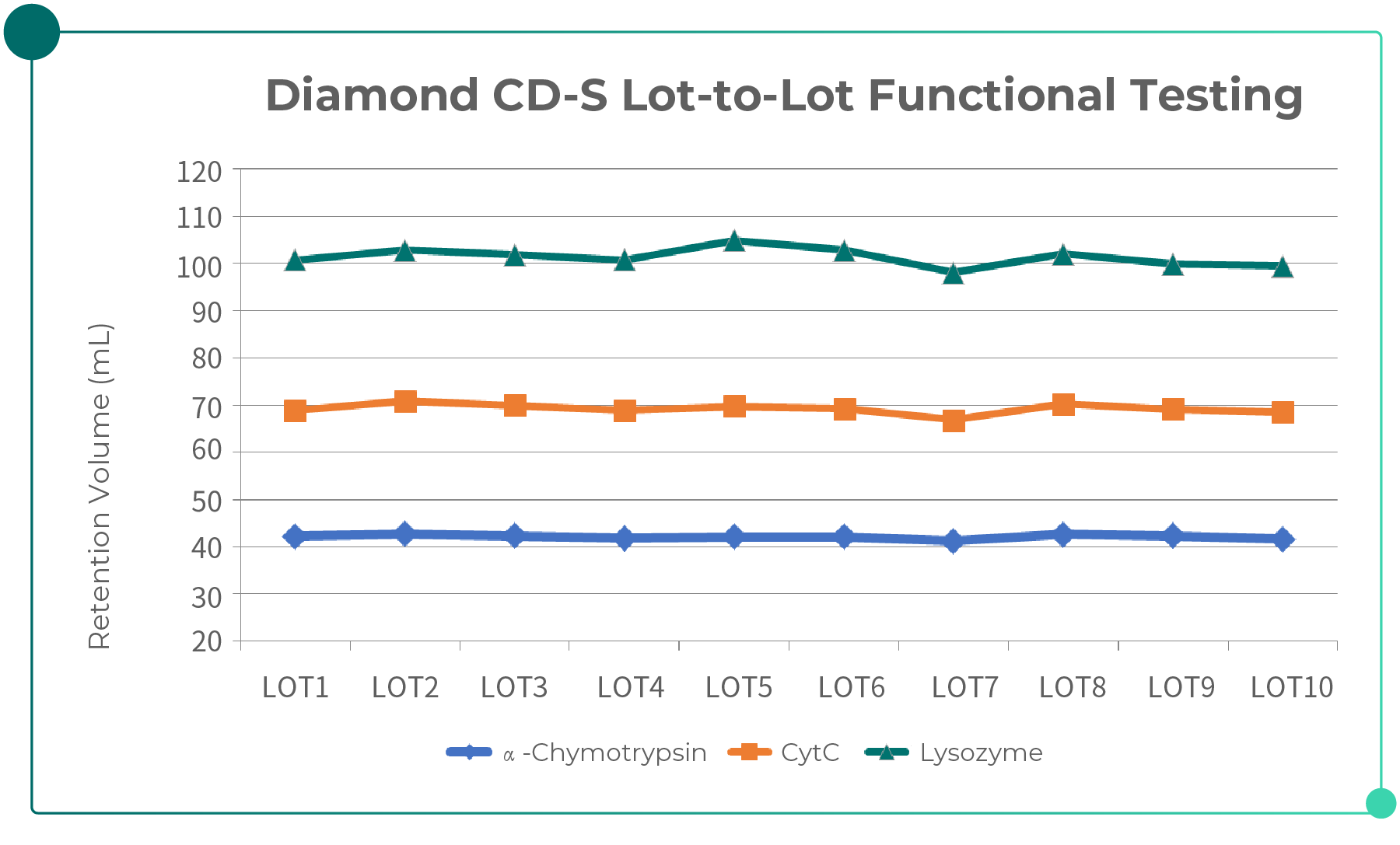
Fig. 3. Lot-to-lot Consistency Data of Bestchrom Chromatography Resins
Supply Chain Reliability and Regulatory Compliance: A Critical Lifeline
Suppliers must operate stable manufacturing and quality systems that comply with applicable regulatory standards, ensuring large-scale, continuous supply with consistent performance across lots. Supply chain security is a critical lifeline for maintaining manufacturing continuity, and becomes particularly important in the face of uncertainties in international trade. Bestchrom has made significant progress in both resin manufacturing capacity and quality, providing biopharmaceutical manufacturers with a broader range of options for resin selection.
• Comprehensive regulatory support documentation → Robust quality management systems
• Strict change control → Avoidance of unnecessary process revalidation
• Development of a secondary source → Mitigation of supply disruption risks (geopolitics/capacity)
In large-scale manufacturing, the selection of chromatography resins goes far beyond identifying the option with the highest binding capacity. Instead, it represents a systems-level decision that requires comprehensive consideration of technical performance, economic cost, supply chain stability, and regulatory compliance.

Conclusion
The selection of industrial-grade chromatography resins is a complex, multi-objective decision-making process that requires achieving an optimal balance among separation performance, process robustness, economic cost, and supply chain security. A successful resin selection strategy involves addressing the requirements of large-scale manufacturing early in process development, selecting platform-based products with high scalability, and ensuring—through systematic process characterization and validation—that biopharmaceutical processes can transition smoothly from the laboratory to commercial production.
Future decision-makers must strike an optimal balance among performance, cost efficiency, process robustness, regulatory compliance, and supply chain considerations. As Bestchrom continues to advance its resin technologies and expand its product portfolio, Bestchrom is well positioned to better support pharmaceutical manufacturers in resin selection. Bestchrom remains committed to serving both Chinese and global biopharmaceutical companies, helping to safeguard the reliable production of compliant, high-quality medicines.









.png)


