Pre-activated Chromatography Resins
Affinity chromatography, characterized by its high specificity, sensitivity, and rapid, efficient performance, is widely applied across biopharmaceuticals, biochemistry, molecular biology, and clinical diagnostics. However, commercially available resin do not always fully meet the requirements of specific projects, making in-house preparation of affinity resins a practical and effective solution.
Pre-activated chromatography resins can be coupled with a specific ligand to generate affinity resin capable of purifying one or multiple target molecules. For example, antibodies, antigens, enzymes, receptors, small nucleic acids, or peptides can be used as ligands to bind their corresponding targets.
Resin Overview

Coupling Protocol
• Coupling Solution A:1mM HCl
• Coupling Solution B:0.1M NaHCO3、0.5M NaCl,pH 8.3
◉ Resin Buffer Exchange
Transfer a given volume of resin into a fritted funnel and wash with pre-cooled Coupling Solution A (0–4 °C) for at least 30 minutes. Use approximately 20 mL of Coupling Solution A per 1 mL of resin.
Note: NHS-activated ligands hydrolyze rapidly at high pH.
◉ Ligand Buffer Exchange and Coupling Reaction
Dissolve the ligand in Coupling Solution B, or exchange it into Coupling Solution B using a Bestdex G-25 column (recommended coupling concentration: 5–10 mg ligand per mL resin). Dilute the washed resin with Coupling Solution A (approximately 0.5 mL Coupling Solution A per 1 mL resin), then mix with an equal volume of the ligand solution. Mix gently and allow to stand for 4 h at room temperature or overnight at 4 °C (do not use a magnetic stirrer).
• Note: The coupling reaction is typically rapid at room temperature. Therefore, optimizing the coupling time is important to preserve ligand bioactivity (prepare the ligand solution prior to resin buffer exchange).
◉ Blocking and Washing
• Blocking: Remove the coupling supernatant and add blocking solution (0.1 M Tris-HCl, pH 8.5). Block for 2–4 h at room temperature.
• Washing: Alternately wash the resin with:
0.1 M Tris-HCl + 0.5 M NaCl, pH 8–9
0.1 M acetate buffer + 0.5 M NaCl, pH 3–4
for 3–6 cycles. Perform each wash using 3 column volumes (CV) of the respective buffer.
• Wash with PBS before use or store in 20% ethanol (the ligand must be stored in 20% ethanol for stability considerations).
Application Examples
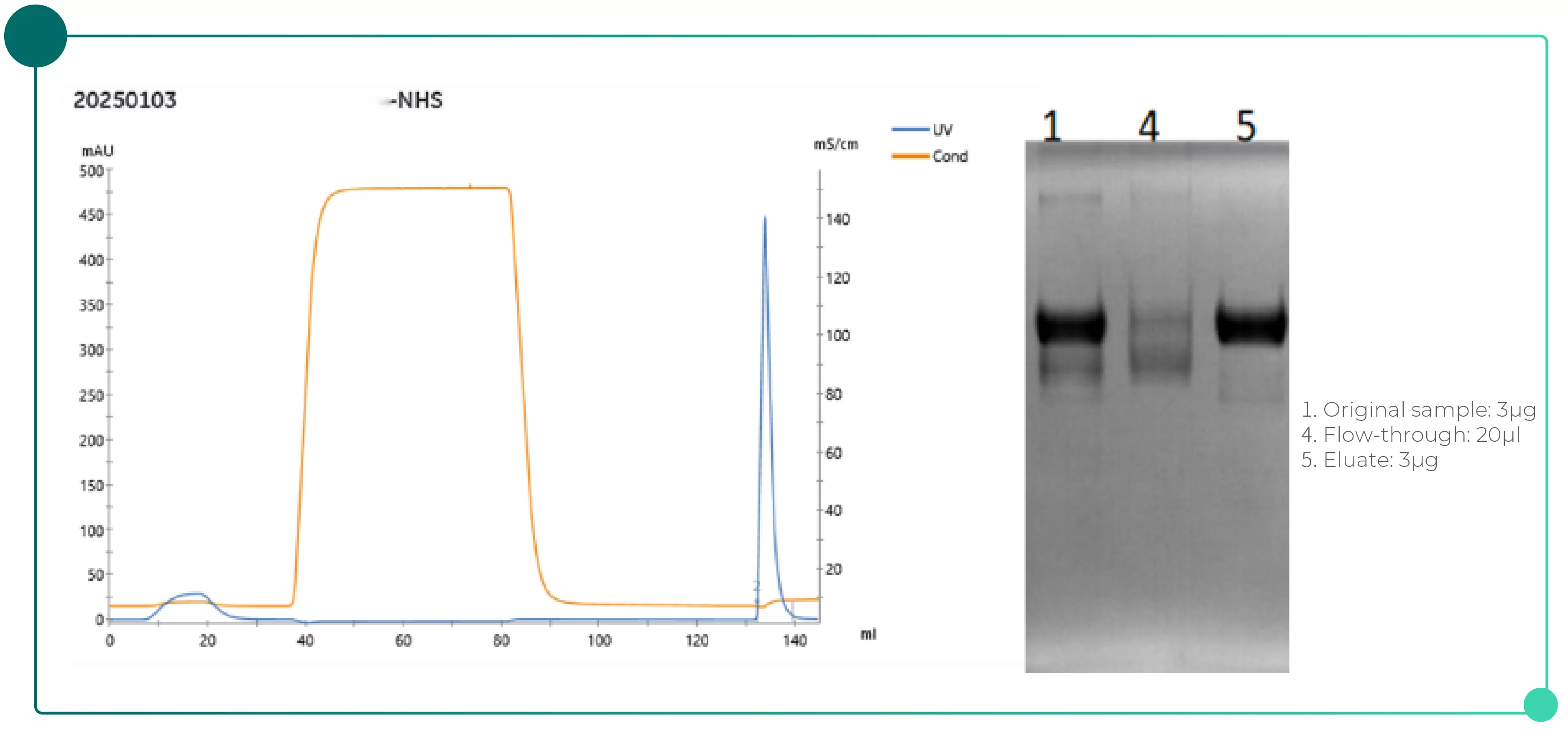
◉ Recombinant protein captured using peptide-coupled NHS-activated Bestarose 4FF
Notes
Generally, when the ligand has a molecular weight < 5 kDa, a spacer arm is recommended. For larger macromolecules, a spacer arm is usually not essential. For example, NHS pre-activated resins have a 10-carbon spacer arm between the reactive site and the matrix, whereas CNBr-activated resins have none. When coupling small-molecule ligands (< 5 kDa), the absence of a spacer arm in CNBr-activated resins may cause steric hindrance that affects binding efficiency. In such cases, NHS-activated resins are strongly recommended. For ligands larger than 5 kDa, the difference between the two is negligible.









.png)


